Abstract
Background
Recurrent mutations in the spliceosome complex are seen in myeloid malignancies, including acute myeloid leukemia (AML), chronic myelomonocytic leukemia (CMML), and myelodysplastic syndromes (MDS). Mutations are detected most frequently in SF3B1, SRSF2, or U2AF1, and are largely mutually exclusive. Recently, a novel SF3B1 modulator, H3B-8800, entered phase-I clinical trials. While the impact of spliceosome mutations at diagnosis and at the time of transplant has been described, there is limited data about the outcomes of patients with splicing factor-mutated disease after the initiation of standard frontline chemotherapies: induction chemotherapy (IC) or hypomethylating agents (HMA).
Methods
Patients with MDS, CMML, or AML with a spliceosome mutation and treated with chemotherapy were identified. Disease risk was assessed using standard criteria (IPSS-R for MDS, ELN for AML, and CPSS for CMML). Overall survival (OS) was defined as the time from start of treatment until death and censored at last known alive. Event free survival (EFS) was defined as the time from start of treatment until relapse, new therapy, or death. We identified the first therapy for newly diagnosed disease (induction chemotherapy, HMA, bone marrow transplant, or other), and grouped remissions as CR (complete remission) or CRi (complete remission with incomplete hematologic recovery). Outcomes were assessed by disease type, mutation, and initial therapy. EFS and OS were estimated by the method of Kaplan and Meier.
Results
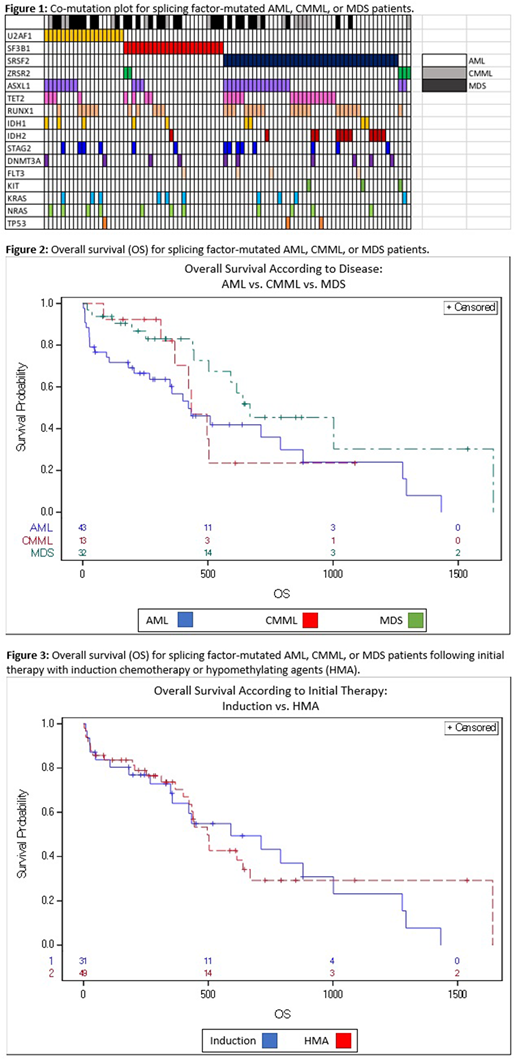
We identified 88 patients diagnosed with AML, CMML, or MDS harboring a mutation in SF3B1 (n=24), SRSF2 (n=43), or U2AF1 (n=21). The median follow-up of the cohort was 637 days. The median age at diagnosis was 69.5 years (33-90), and there was no difference according to mutation (p=0.907). A diagnosis of AML was slightly more common among patients with SRSF2 mutations at the time of treatment, but this was not statistically significant (27/43, p=0.07). Patients with SF3B1 mutations were less likely to have co-mutated ASXL1 (12%, vs 42% with U2AF1 and 40% with SRSF2, p=0.0384). SRSF2 disease more often had co-mutated IDH2 (26%, vs 0% with U2AF1 and 4% with SF3B1, p=0.01). Other co-mutations did not vary significantly between U2AF1, SF3B1, or SRSF2-mutated disease (Figure 1). Patients who had a mutation in SF3B1 had a longer duration from diagnosis to first therapy (median 65 days, range 0-1159) than those with U2AF1 (median 39d) or SRSF2 (median 9d) mutations (p=0.045).
A total of 31 patients received induction chemotherapy, while 50 received HMA as initial therapy; 3 patients were transplanted, and 5 received other chemotherapies (e.g. enasidenib). The remission rate for patients treated with induction was 55%, and there was no difference in rate of remission (CR+CRi) according to mutation type (U2AF1 50%, SF3B1 60%, SRSF2 53%, p=1.00). Patients treated with HMA had a remission rate of 22%, with a trend toward lower remission rates in SF3B1-mutated disease (0/11, vs. U2AF1 20% and SRSF2 33%, p=0.08).
We performed a multivariate analysis for overall survival incorporating mutation type, initial treatment, disease risk group, age at the time of treatment, and disease (MDS, AML, or CMML). Compared to MDS, there was no difference in OS according to whether patients had splicing-factor mutated AML (p=0.45) or CMML (p=0.61) (Figure 2). Interestingly, although CR rates differed between all patients receiving induction or HMA, there was no difference in OS between these same groups (Figure 3, p=0.803).
Conclusion
In this study, there was no difference in response to frontline treatment for SF3B1-mutated myeloid malignancies compared to U2AF1- or SRSF2-mutated disease. Interestingly, there was no difference in survival according to whether patients had splicing-factor mutated AML, MDS, or CMML at the time of treatment; moreover, there was no difference in survival with induction compared to HMA therapy. Together, these point to a rationale to include AML, MDS, and CMML patients together on trials targeting these mutations, and the general need to improve on the poor outcomes with standard induction or HMA therapy.
Amrein:Takeda: Research Funding. Fathi:Celgene: Consultancy, Honoraria, Research Funding; Boston Biomedical: Consultancy, Honoraria; Jazz: Honoraria; Astellas: Honoraria; Agios: Honoraria, Research Funding; Seattle Genetics: Consultancy, Honoraria; Takeda: Consultancy, Honoraria. Brunner:Novartis: Research Funding; Celgene: Consultancy, Research Funding; Takeda: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal